
You can multiply coefficients of reaction with fractions like 3/2.Įxample: Balance following chemical equation.Balancing chemical equations, you should balance H and O after balancing other elements.In standard conditions, 1mol gas is 22,4 liters, number of moles of C In chemical reactions mass is always conserved. Thus, number of molecules is not conserved.Įxample: 4 g substance A reacts with 2,5 g substance B and 1,4 liters C gas and 3,5 g D are produced. In this reaction, 2 molecule H 2 react with 1 molecule O 2 and 2 molecule H 2O is produced. In this reaction 1 H 2 molecule and 1Cl 2 molecule reacts and 2 molecule HCl is produced. In a chemical reaction, number of molecules is not conserved always.Total charges of reactants are equal to total charges of products. 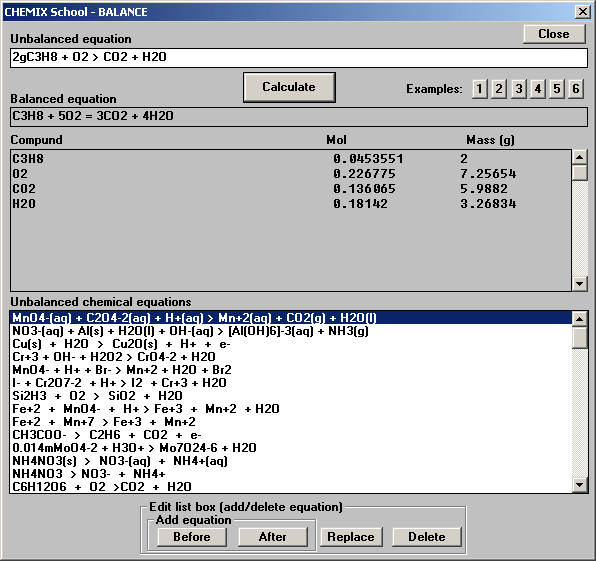 
Charges of elements/compounds are conserved.Masses of reactants are equal to masses of products. In other words, sum of the atoms in reactants part is equal to sum of the atoms of products. Number of atoms of elements are conserved.In a chemical reaction, mass is conserved, it is not lost or created. Balancing Chemical Reactions with Examples
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
